The bitter truth about depression: How astringent flavanols “wake up” the brain and restore mood naturally
05/07/2026 / By Lance D Johnson

For millions trapped in the fog of depression, the standard pharmaceutical path offers little more than a roll of the dice. Selective serotonin reuptake inhibitors fail roughly half the people who try them. Side effects like sexual dysfunction, insomnia, and emotional numbness drive countless patients to abandon treatment altogether. The World Health Organization now projects that depression will become the leading cause of disability by 2030, a crisis that accelerated by 25% during the COVID-19 tyranny. Against this backdrop, a growing body of research is pointing toward an unexpected ally in the fight against mood disorders: the very compounds that make dark chocolate, red wine, and green tea taste slightly bitter and drying on the tongue.
A new preclinical study from Japan’s Shibaura Institute of Technology, published in Current Research in Food Science, suggests that the astringent sensation caused by flavanols may act as a direct signal to the brain’s alertness center, triggering effects remarkably similar to a mild workout for the nervous system. Meanwhile, a comprehensive review published in Frontiers in Pharmacology examining decades of flavonoid research has documented antidepressant properties across multiple sub-classes of these plant compounds, with mechanisms that include neurotransmitter modulation, anti-inflammatory activity, and promotion of brain-derived neurotrophic factor expression. The convergence of these findings raises a compelling question: could the bitter taste of certain foods be part of nature’s own antidepressant delivery system?
Key points:
- Astringent flavanols activate the locus coeruleus-noradrenaline system, the brain’s alertness center, within minutes of oral administration in mice
- Flavonoids from multiple subclasses demonstrate antidepressant effects through neurotransmitter modulation, BDNF enhancement, and anti-inflammatory pathways
- The “sensory nutrition” concept suggests that flavor itself, not just absorbed nutrients, may trigger physiological benefits
- Only a small fraction of flavanols enter the bloodstream, yet their effects on brain function are profound
- Citrus bioflavonoids like hesperidin and naringenin show particular promise for mood regulation
The flavanol puzzle: Why low absorption doesn’t mean low impact
For years, scientists have been puzzled by a curious contradiction. Flavanols, a subtype of polyphenols found abundantly in dark chocolate, berries, red wine, and tea, have been linked to improvements in cognitive performance, cardiovascular health, and even protection against age-related brain decline. Yet when researchers measured how much of these compounds actually made it into the bloodstream after digestion, the numbers were strikingly low. Something didn’t add up.
The team at Shibaura Institute of Technology, led by researcher Yasuyuki Fujii, decided to investigate this paradox directly. They gave 10-week-old mice oral doses of flavanols at 25 or 50 milligrams per kilogram of body weight and observed what happened. The results were striking. The flavanol-treated mice showed increased physical activity and exploration behavior, improved learning and memory performance, and most significantly, activation of the locus coeruleus-noradrenaline system, essentially the brain’s alarm and alertness center.
Using mass imaging and in situ hybridization analyses, the researchers detected high-intensity noradrenaline originating from the locus coeruleus in the hypothalamus and brainstem immediately after flavanol administration. The mice’s brains responded as if they had received a mild workout, triggering a cascade of neurotransmitters including dopamine and norepinephrine, while also activating stress-response pathways like the sympathetic-adrenal-medullary axis and the hypothalamic-pituitary-adrenal axis.
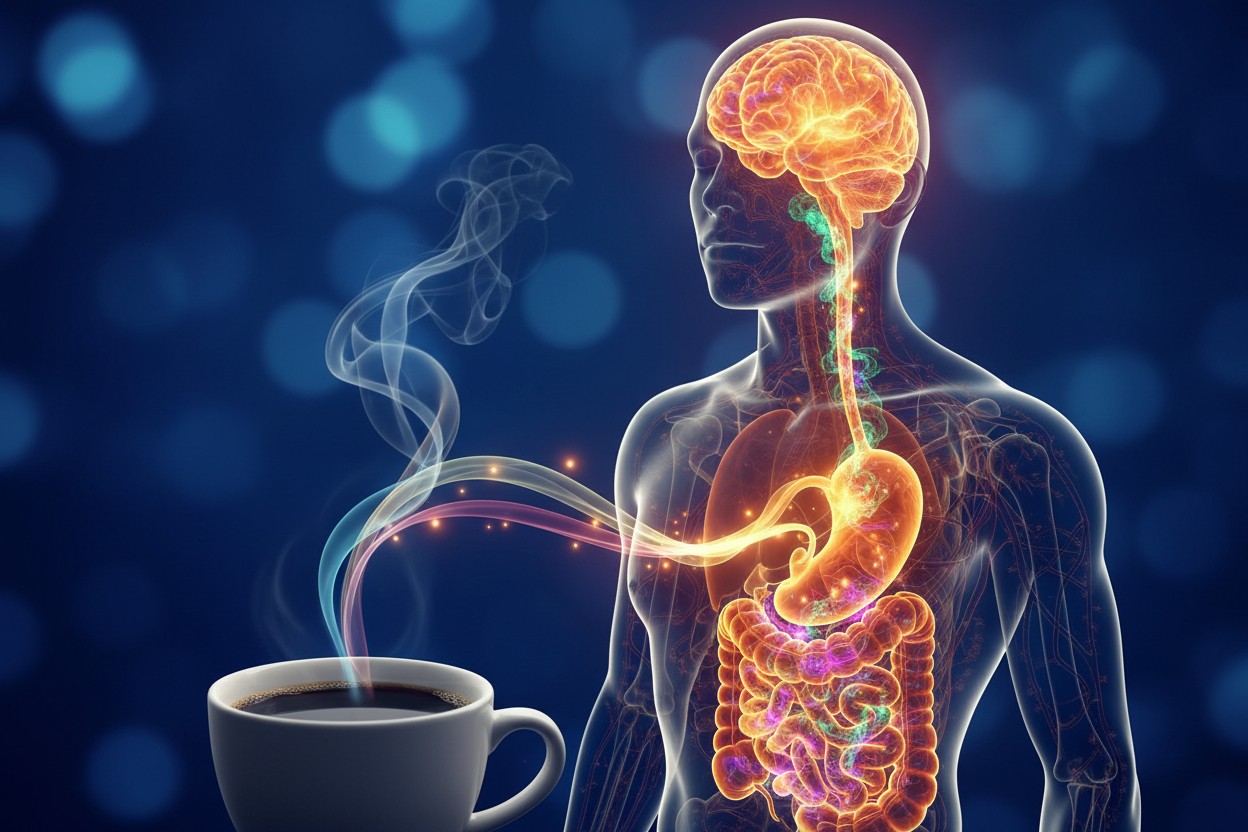
The researchers propose that this effect is not primarily about flavanols crossing the blood-brain barrier. Instead, the astringent taste itself may be the trigger. The mouth-puckering sensation appears to send signals directly to the brain via sensory nerves, a concept they call “sensory nutrition.” In their own words, the findings “highlight how astringent stimulants FLs may activate brain function and the autonomic nervous system via gastrointestinal stimulation, causing physiological changes.” They concluded that “the sensory properties of food are important for maintaining homeostasis and promoting human health.”
This represents a paradigm shift in nutritional science. The prevailing assumption has been that the health benefits of food depend almost entirely on what gets absorbed into the bloodstream. But this research suggests that the simple act of tasting certain compounds could prime the brain, activate alertness pathways, and shape physiological responses in real time, independent of absorption.
The flavonoid antidepressant arsenal: From citrus peels to cocoa beans
While the flavanol study opens a new window into how taste may influence mood, a separate comprehensive review published in Frontiers in Pharmacology by Alizadeh and colleagues documents a vast body of evidence showing that flavonoids from multiple sub-classes possess genuine antidepressant properties through well-established biological mechanisms.
The review, which examined studies up to July 2022, catalogued antidepressant effects across chalcones, anthocyanins, flavonols, isoflavonoids, flavones, and flavanones. Each subclass appears to work through multiple pathways, including modulation of neurotransmitter systems, enhancement of brain-derived neurotrophic factor, reduction of oxidative stress, and suppression of neuroinflammation.
Among the most promising compounds is hesperidin, a citrus bioflavonoid found abundantly in oranges, lemons, and other citrus fruits. Studies have shown that hesperidin reduces levels of pro-inflammatory cytokines like IL-6, IL-1?, and TNF-? in animal models of depression, while also increasing BDNF expression and modulating serotonin and dopamine levels. The compound appears to work through multiple mechanisms simultaneously, including inhibition of the L-arginine-NO-cGMP pathway, activation of the Nrf2/ARE antioxidant pathway, and even interaction with k-opioid receptors.
Naringenin, another citrus flavanone found in grapefruit peels, has demonstrated antidepressant effects through restoration of the kynurenine pathway, a metabolic pathway of tryptophan whose dysregulation is increasingly recognized as a key factor in depression. Naringenin also activates the Sonic Hedgehog signaling pathway, upregulates BDNF expression, and enhances serotonergic and noradrenergic systems. Its antioxidant and anti-inflammatory properties further contribute to its therapeutic potential.
The review documented that flavonoids from other sub-classes show similar promise. Quercetin, abundant in apples, onions, and green tea, modulates inflammation, inhibits MAO-A activity, and protects against oxidative stress. Genistein, an isoflavonoid from soy, down-regulates microRNAs linked to depression and regulates serotonergic enzymes. Apigenin, found in grapefruits, oranges, and celery, inhibits inflammatory cytokines and increases autophagy through the AMPK/mTOR pathway.
What makes these findings particularly significant is the side effect profile. Unlike conventional antidepressants, which frequently cause sexual dysfunction, insomnia, weight gain, and emotional blunting, flavonoids appear to produce minimal adverse effects at dietary doses. The review noted that “there were no severe side effects” across the studies examined, though it cautioned that high-dose supplements could potentially cause toxicity.
Flavanol and flavonoid list with antidepressant mechanisms and food sources
From the study, the following compounds demonstrated antidepressant or mood-related properties:
1. Chalcones
- Phloridzin: Enhances expression of GSH, BDNF, TrkB, CREB, and ERK. Found in apple peels.
- DHIPC: Increases serotonin, noradrenaline, and 5-HIAA in the hippocampus.
2. Anthocyanins
- Cyanidin: Inhibits monoamine oxidase activity, increases BDNF production, upregulates PI3K/AKT/FoxG1/FGF-2 pathway, decreases pro-inflammatory cytokines. Found in purple cauliflower, pomegranate, blueberries.
- Delphinidin: Inhibits oxidative stress. Found in maqui berries, blueberries, grapes.
- Malvidin: Maintains synaptic plasticity by increasing Rac1 expression. Found in red wine, blueberries.
3. Flavonols
- Rutin: Increases noradrenaline and serotonin in synaptic cleft, protects from oxidative stress. Found in citrus fruits, buckwheat, apples.
- Kaempferol: Enhances anti-inflammatory and antioxidant effects via AKT/?-catenin cascade, increases NE, DA, and 5-HT. Found in tea, broccoli, tomatoes, grapes, Ginkgo biloba.
- Kaempferol-3-O-D-glucoside: Increases NE, DA, and 5-HT. Found in Apocynum venetum leaves.
- Kaempferitrin: Regulates serotonergic system via 5-HT1A receptors, regulates HPA axis. Found in Justicia spicigera plants.
- Myricetin: Improves glutathione peroxidase activity in hippocampus, reduces plasma corticosterone, normalizes BDNF levels. Found in berries, nuts, red wine, tea.
- Myricitrin: Inhibits nitric oxide, promotes hippocampal neurogenesis. Found in berries.
- Morin: Elevates epinephrine, norepinephrine, and serotonin; decreases inflammatory markers TNF-alpha, TLR-4, NLRP3, IL-1beta. Found in figs, almonds, red wine.
- Quercetin: Modulates inflammation, inhibits MAO-A, prevents oxidative stress, increases BDNF, increases NE, DA, and 5-HT. Found in apples, onions, broccoli, green tea, Ginkgo biloba.
- Quercetin-3-O-D-glucoside: Increases NE, DA, and 5-HT. Found in Apocynum venetum leaves.
- Icariin: Anti-oxidant, inhibits NF-kB signaling and NLRP3 inflammasome, increases BDNF, regulates HPA axis, increases monoamine neurotransmitters. Found in Herba epimedii.
- Hyperoside: Activates D2 dopamine receptors, inhibits NLRP1 inflammasome, increases BDNF and CREB expression, regulates HPA axis. Found in Hypericum perforatum (St. John’s wort).
- Fisetin: Regulates serotonin and noradrenaline levels, reverses pro-inflammatory cytokine overexpression, activates TrkB signaling, downregulates TNF-?/NLRP3. Found in strawberries, apples, persimmons.
4. Isoflavonoids
- Genistein: Down-regulates miR-221/222 targeting connexin 43, regulates serotonergic enzymes. Found in soybeans, legumes.
- Daidzein: Decreases stress-related hormones, mitigates HPA axis hyperactivity, decreases inflammatory cytokines. Found in soybeans, legumes.
5. Flavones
- Apigenin: Inhibits inflammatory cytokines, iNOS, and COX-2; regulates dopaminergic mechanisms; increases autophagy through AMPK/mTOR pathway. Found in grapefruits, oranges, celery, onions.
- Baicalein: Reverses reduction of ERK phosphorylation, enhances hippocampal BDNF expression, promotes neurogenesis via cAMP/PKA pathway, ameliorates neuroinflammation. Found in Scutellaria baicalensis (Chinese skullcap).
- Chrysin: Enhances BDNF expression, increases Na+,K+-ATPase activity, decreases ACTH and corticosterone, inhibits inflammatory cytokines, interacts with GABA receptors. Found in propolis, honey, passionflowers.
- Luteolin: Lowers corticosterone and ACTH, inhibits MAO enzyme, decreases norepinephrine while increasing serotonin, enhances synapsin levels, inhibits hippocampal inflammation. Found in carrots, cabbage, parsley, broccoli, celery, apple peels.
- Nobiletin: Reverses neuroinflammation, promotes autophagy, suppresses NLRP3 inflammasome via AMPK pathway, interacts with noradrenergic, serotonergic, and dopaminergic systems. Found in citrus peels.
- 7,8-Dihydroxyflavone: Elevates BDNF expression, acts as TrkB receptor agonist, modulates nitric oxide signaling. Found in Godmania aesculifolia, primula plants.
- Amentoflavone: Interacts with 5-HT2 receptors, ?1- and ?2-adrenoceptors. Found in Ginkgo biloba, Hypericum perforatum.
6. Flavanones
- Hesperidin: Reduces inflammatory cytokines, suppresses microglia activation, inhibits L-arginine-NO-cGMP pathway, increases hippocampal BDNF, interacts with k-opioid receptors and 5-HT1A receptors, activates Nrf2/ARE pathway, inhibits acetylcholinesterase. Found in citrus fruits, especially fingered citron.
- Naringenin: Restores kynurenine pathway, upregulates SHH signaling, increases BDNF expression, inhibits inflammatory cytokines, modulates NF-kB, suppresses microglia activation, involves serotonergic and noradrenergic systems. Found in grapefruit peels, citrus fruits.
- Naringin: Reduces oxidative damage, lowers plasma corticosterone, modulates 5-HT1A and kappa-opioid receptors, inhibits NMDA receptors, activates PKA/CREB/BDNF pathway, promotes neurogenesis, inhibits AChE activities. Found in grapefruit, citrus fruits.
- Eriodictyol: Improves cognitive impairments induced by chronic stress. Found in citrus fruits, medicinal plants.
The convergence of these two research streams, the sensory nutrition paradigm from the flavanol study and the extensive mechanistic evidence from the flavonoid review, suggests that diet may play a far more sophisticated role in mental health than previously appreciated. The fact that flavonoids work through multiple pathways, neurotransmitter modulation, BDNF enhancement, anti-inflammatory effects, and oxidative stress reduction, means they may address the biological complexity of depression more comprehensively than single-target pharmaceutical drugs.
Sources include:
Submit a correction >>
Tagged Under:
anthocyanins, Apigenin, BDNF, citrus bioflavonoids, depression treatment, dietary flavonoids, flavanols, gut-brain axis, Hesperidin, mood regulation, naringenin, natural antidepressants, neuroinflammation, noradrenaline, phytonutrients, polyphenols, quercetin, research, sensory nutrition
This article may contain statements that reflect the opinion of the author